The sustainable transition toward FBS alternatives: Greening cell culture
Blog by Helena (Huiyuan) Meng, third year PhD candidate, School of Biological sciences
Trigger warning: This article discusses foetal bovine serum (FBS) sourcing and includes references to animal slaughter and animal welfare impacts.
Have you ever stopped to wonder where foetal bovine serum (FBS) comes from when it’s added to cell culture media? For more than six decades, FBS has been a staple in labs, valued for its rich mix of proteins, growth factors, fatty acids and vitamins that support the growth of mammalian cells (Lee et al., 2022).
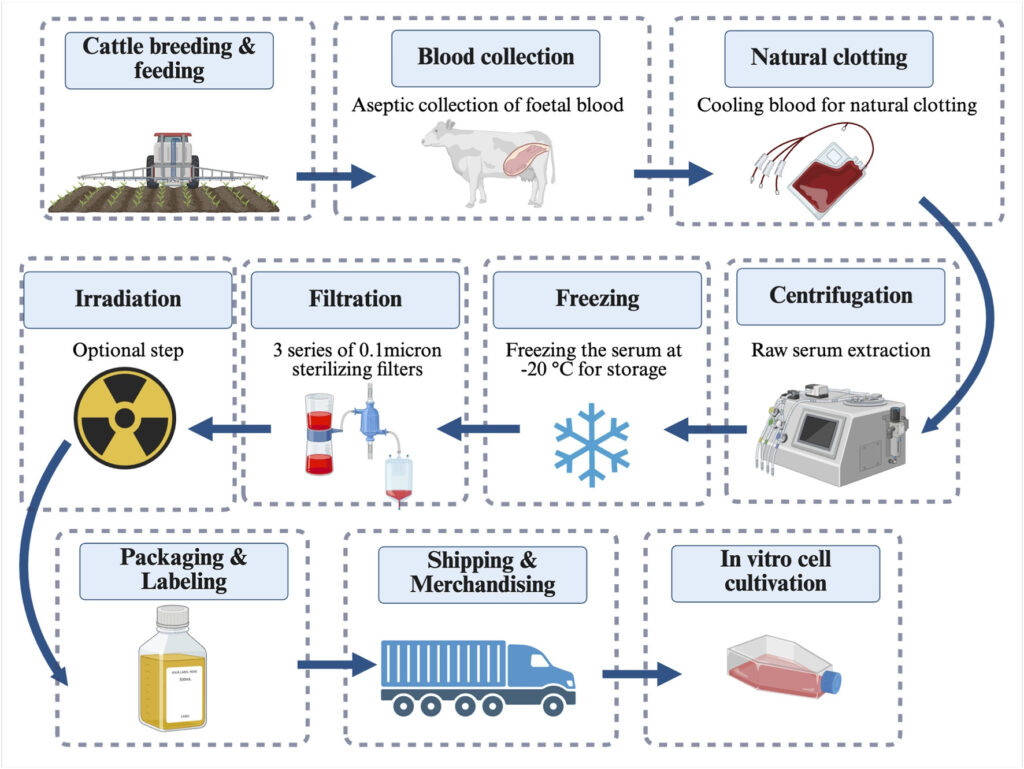
FBS production process. Meng and Day (2026)
But behind this routine practice lies broader concerns around sustainability, cost, and animal welfare. FBS is obtained from bovine foetuses during the slaughter of pregnant cows (Liu et al., 2023). As global demand continues to rise, growing by an estimated 5–7% each year, production is estimated to slaughter over one million foetal calves annually (Weber et al., 2025).
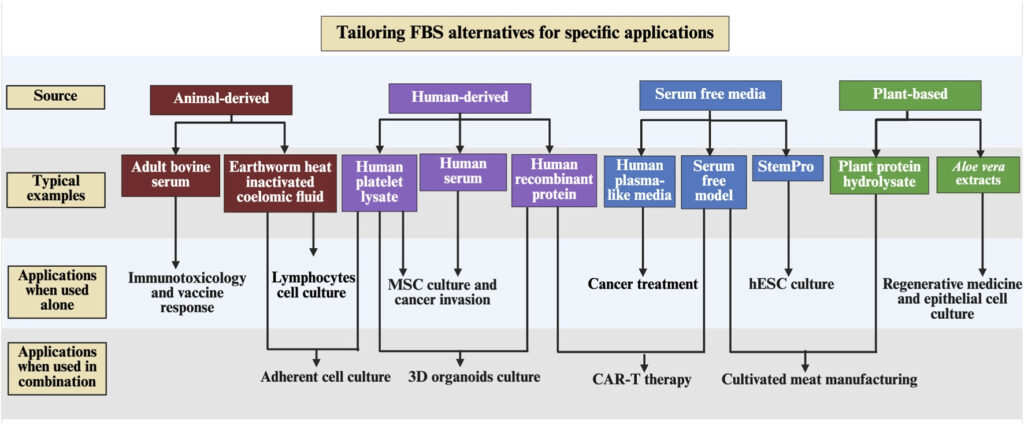
In response, scientists are working to develop FBS alternatives, including human and animal derived, plant-based supplements, and chemically defined serum-free media, aiming to make research not only effective but also more environmentally sustainable and ethical (Weber et al., 2025).
The transition towards FBS alternatives
In our review paper, we explored the applicability of FBS alternatives across different research areas, and proposed guidance for tailoring these alternatives to better support the transition away from traditional FBS use (Meng and Day, 2026).
This shift in cell culture practices is closely aligned with UK government directives aimed at reducing the use of animal-derived products in research, as well as with the University ongoing UKRI SPARKHub initiatives focused on sustainability and innovation. It also contributes to minimizing the environmental impact associated with livestock farming, including resource consumption and greenhouse gas emissions.
In parallel, resources such as the Foetal Calf Serum-free Database established by the 3Rs Centre Utrecht, together with advances in cultured meat technologies and growing institutional incentives, are accelerating the development, dissemination, and adoption of FBS alternatives across scientific research, biomanufacturing, and clinical applications (3Rs Centre Utrecht, 2024).
A call for sustainable research
As an early-career researcher, working on this review has helped me to think more critically about the hidden costs behind routine laboratory practices. I am inspired to contribute to the development and adoption of more sustainable, ethical and application-specific culture system, and to advocate for research where transparency, reproducibility, and ethical sourcing are no longer optional, but fundamental expectations.
Additional information:
This work aligns closely with the United Nations Sustainable Development Goals, particularly Goal 12 (Responsible Consumption and Production) and Goal 9 (Industry, Innovation and Infrastructure).
Please check our review paper: doi: 10.3389/ftox.2026.1776815
Please check the Foetal Calf Serum-free Database: https://fcs-free.org/fcs-database
References:
3Rs Centre Utrecht (2024). Foetal calf serum (FCS)-free database RRID:SCR_018769. Available online at: https://fcs-free.sites.uu.nl/database/ (Accessed April 23, 2026).
Lee, D. Y., Lee, S. Y., Yun, S. H., Jeong, J. W., Kim, J. H., Kim, H. W., et al. (2022). Review of the Current research on foetal bovine serum and the development of cultured meat. Food Sci. Animal Resour. 42, 775–799. doi:10.5851/kosfa.2022.e46.
Liu, S., Yang, W., Li, Y. and Sun, C. (2023). Fetal bovine serum, an important factor affecting the reproducibility of cell experiments. Scientific Reports, 13, 1942. doi:10.1038/s41598-023-29060-7.
Meng, H. and Day, PJR. (2026) Exploring ethical, sustainable and effective foetal bovine serum alternatives for in vitro mammalian cell culture. Front. Toxicol. 8:1776815. doi:10.3389/ftox.2026.1776815.
Weber, T., Malakpour-Permlid, A., Chary, A., D’Alessandro, V., Haut, L., Seufert, S.,et al. (2025). Foetal bovine serum: how to leave it behind in the pursuit of more reliable science. Front. Toxicol. 7, 1612903. doi:10.3389/ftox.2025.1612903.

0 Comments